
Lead acid batteries have been critical to powering vehicles since the 19th century. Relatively inexpensive, these batteries provide a large amount of current instantaneously. This is how the battery works:
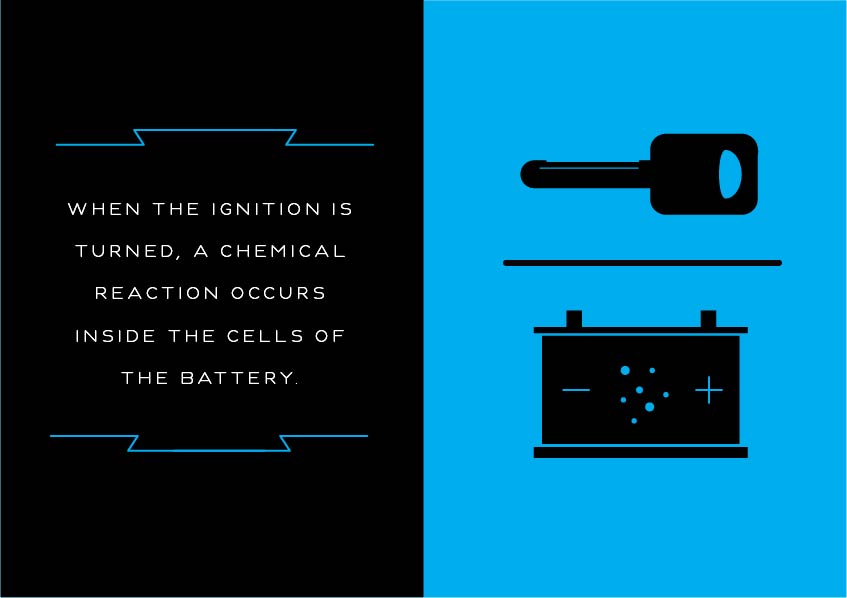
The battery consists of a set of cells which have lead plates immersed in concentrated sulfuric acid and water. This mixture functions as an electrolyte, which conducts electricity. When the ignition is turned, a chemical reaction occurs inside the cells of the battery.
Then, the battery’s chemical energy is converted to electrical energy — a large current — which kicks the electric starter motor into action. The starter motor converts the electric current into mechanical energy, rotating the internal combustion engine so it can then operate under its own power.
After it helps start the vehicle, the battery provides power to accessory items, such as windows, windshield wipers, the motors that operate the windows, locks, and radio.
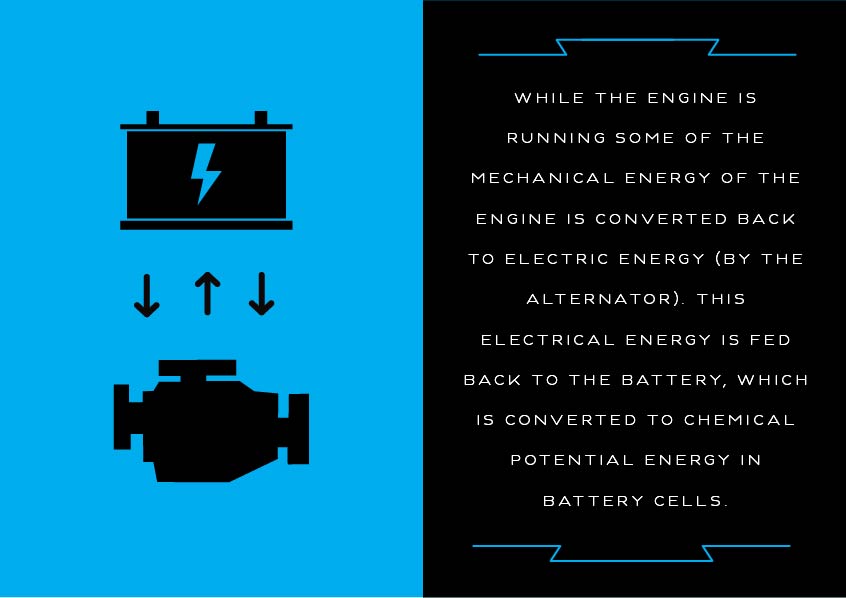
With the starting of the engine and as you operate vehicle accessories, you are draining energy from the battery. So, while the engine is running, some of the mechanical energy of the engine is converted back to electric energy (by the alternator). This electrical energy is fed back to the battery, which is converted to chemical potential energy in battery cells.
Keep in mind: The standard lead acid battery is potentially dangerous because it contains sulfuric acid, and a maltreated battery (improperly charged or recharged) can cause the formation of explosive hydrogen gas. Modern car batteries are vented to prevent this problem, but it’s important you follow any manufacturer’s directions for installing or recharging a battery.
- Prior to using or installing any of these products always consult your vehicle’s owner’s manual for compatibility and warranty information.
- Always take appropriate safety precautions when working on or operating your vehicle. Take the necessary steps to help prevent injuries; always use protective gear like helmets, safety goggles, and gloves.


Don't miss out on new content
Thanks for signing up. Set your password and start earning reward points for everything you do on the site.
You already have a Team Valvoline account. Sign in here.
Did you forget your password?